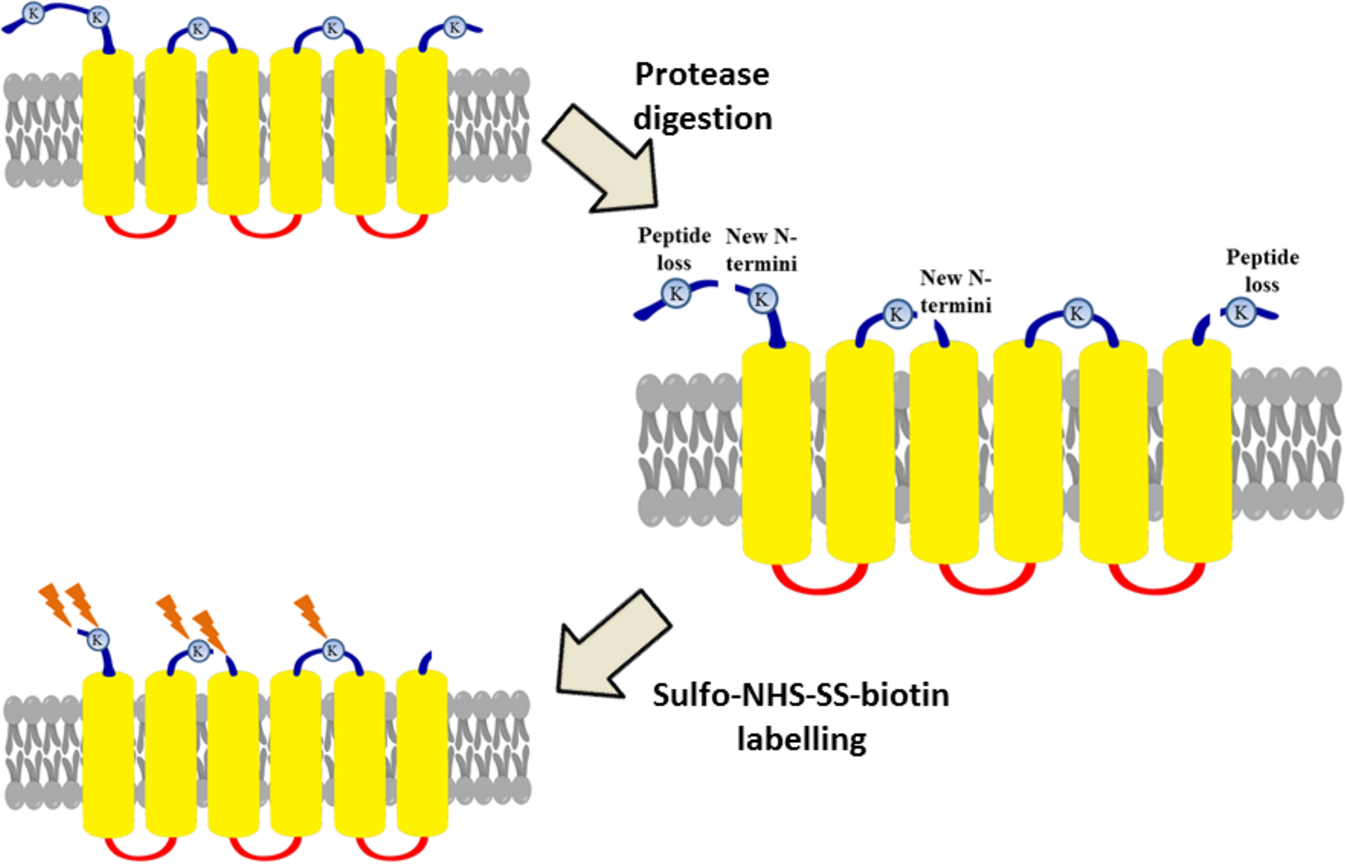
Partial proteolysis improves the identification of the extracellular segments of transmembrane proteins by surface biotinylation | Scientific Reports

Proximity biotinylation provides insight into the molecular composition of focal adhesions at the nanometer scale | Science Signaling
Proteomic Analysis of the Ubiquitin Landscape in the Drosophila Embryonic Nervous System and the Adult Photoreceptor Cells | PLOS ONE
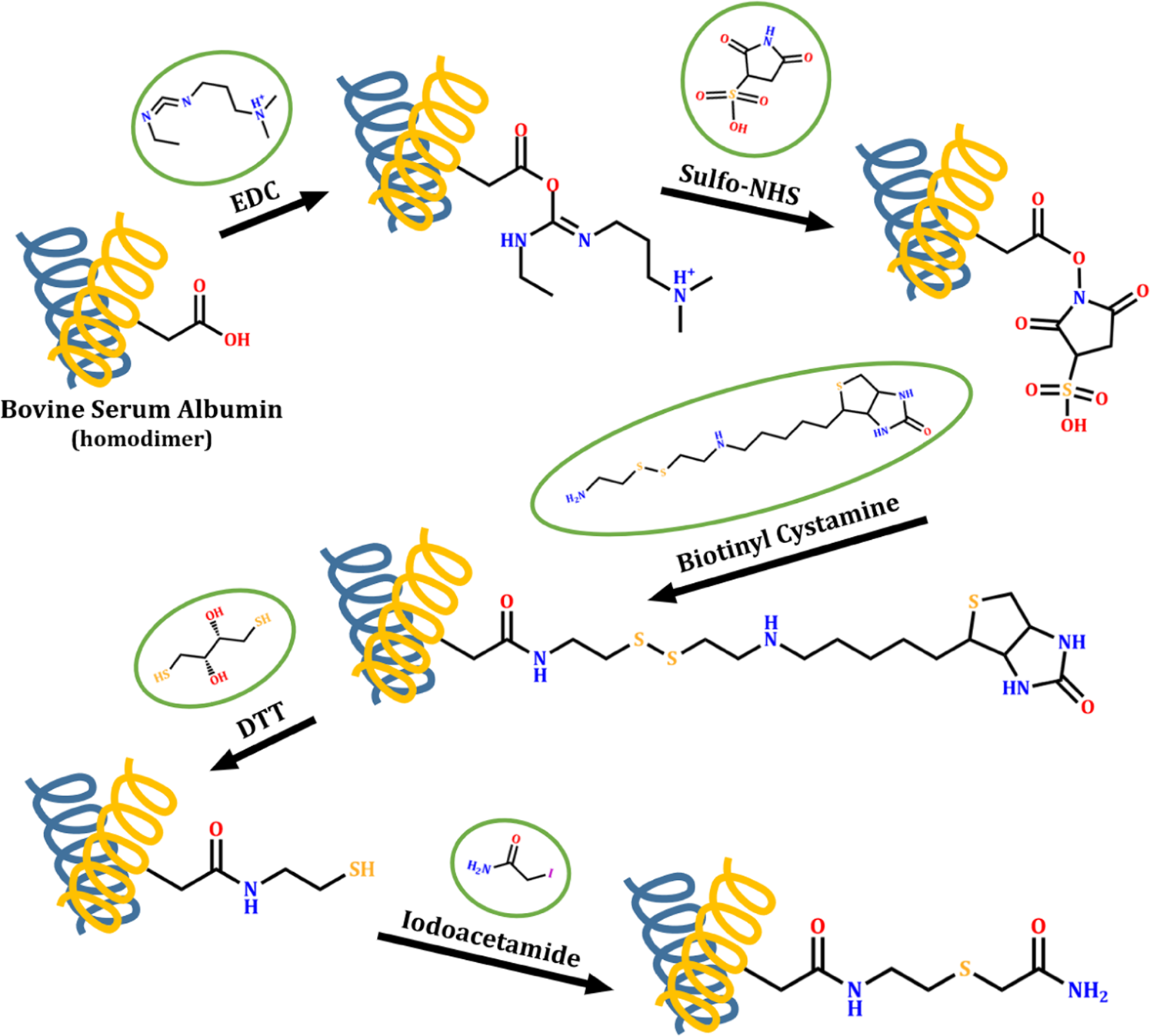
Covalently modified carboxyl side chains on cell surface leads to a novel method toward topology analysis of transmembrane proteins | Scientific Reports
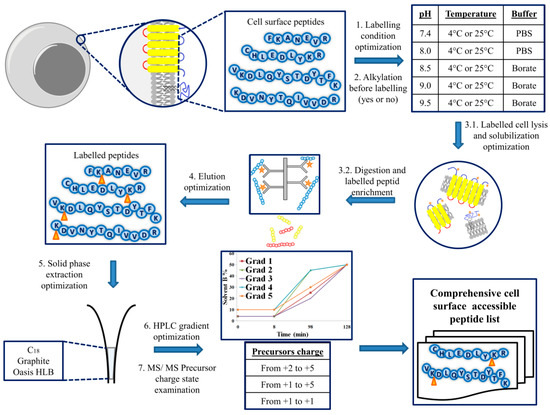
IJMS | Free Full-Text | Comprehensive Discovery of the Accessible Primary Amino Group-Containing Segments from Cell Surface Proteins by Fine-Tuning a High-Throughput Biotinylation Method

Purification scheme, elements and results for producing biotin-labeled... | Download Scientific Diagram

Split‐BioID: a proximity biotinylation assay for dimerization‐dependent protein interactions - De Munter - 2017 - FEBS Letters - Wiley Online Library

IJMS | Free Full-Text | Comprehensive Discovery of the Accessible Primary Amino Group-Containing Segments from Cell Surface Proteins by Fine-Tuning a High-Throughput Biotinylation Method

Assessment of Streptavidin Bead Binding Capacity to Improve Quality of Streptavidin-based Enrichment Studies | Journal of Proteome Research